Medical Additive Manufacturing
Medical Additive Manufacturing
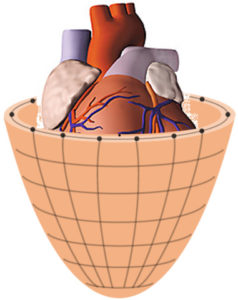

| left: Schematic diagram of the heart actor structure for the treatment of cardiac insufficiency in the rapid prototyping procedure, right: SLS casting mold with complex structures, defined mechanical properties and lost core |
Plastics offer a high adjustability of chemical, physical and mechanical properties and provide an increased potential for application in the medical sector. Because of plastics material properties, bio-resorbable polymers, for example, can be used as a carrier for a dynamically controlled drug delivery system. The use of plastics in implants and prostheses thus enables local and time-defined medication dispensing.
Currently, additive manufacturing processing with plastics in the medical field is mainly restricted to the area of modeling and for extracorporeal applications due to the limited range of biocompatible materials. Therefore, new methods and materials for the production of individual implants for plastic surgery and “tissues” for tissue replacements are to be explored. The main advantage is the production of patient-customized implants which can be directly derived from data, e.g. from a computer tomography scan of the patient. Current research deals with the possibility of directly and indirectly manufacturing implants. With the direct manufacturing method, the implant is directly built by an additive manufacturing method and is thus limited in its material spectrum, depending on the additive manufacturing method. The indirect manufacturing is carried out by first additively manufacturing a mold which can subsequently be used for casting the implant. This method affords the use of any medical-grade casting polymer. With this rapid tooling process, it is possible to mold flexible implant structures for organ implants.
Current research fields at the LKT in the field of additive manufacturing processing (e.g. the selective laser melting of polymers) is both material and processing optimization in order to develop a potentially wider application basis, both for medical and direct manufacturing processes.
